The Problem
Adults who undergo surgical heart procedures and infants with congenital heart defects face the risk of bacterial endocarditis (BE). This infection develops when bacteria colonize heart valves and other structures – and is generally fatal without immediate and long-term antibiotic therapy.
Early detection is vital, and current treatment strategies should be optimized to combat the rising threat of antibiotic resistant bacteria. Azad Eshghi, a postdoctoral fellow at U of T, earned a Ted Rogers Centre for Heart Research Education Fund award to test novel methods of treating BE.
The Concept
The flow of blood through our veins exerts a force that bacteria must counter to successfully attach to the inner lining of veins. Eshghi, guided by supervisors at UHN and U of T, aims to identify new molecules that will inhibit bacteria from attaching to heart valve endothelial lining. But to be accurate, that rush of blood must be recreated.
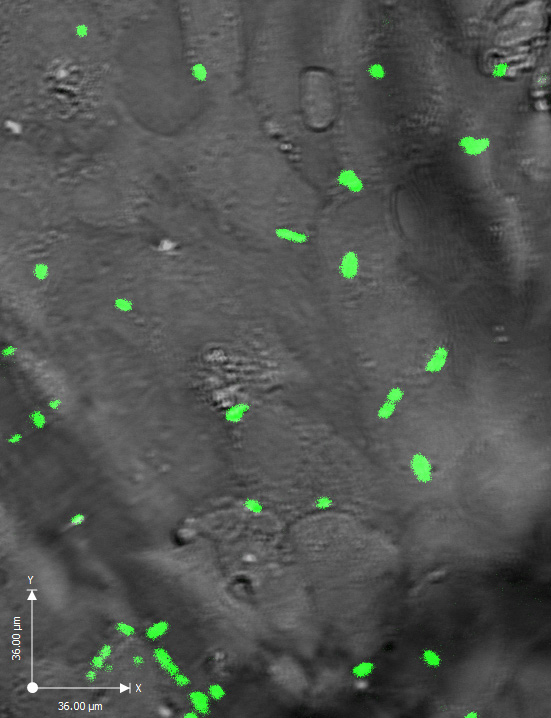
“If you test inhibitors under static conditions you aren’t mimicking the conditions of the heart,” Eshghi says. “We are establishing a system where we create a fluid shear force that bacteria would actually experience in the heart.”
This novel idea involves microplates that have 96 separate wells that can be subjected to fluid shear force (to mimic blood flow). Researchers coat these wells with cardiac valve cells, and introduce compounds that would potentially detach bacteria. The plan is to screen 5,000 compounds to seek those that inhibit bacteria from attaching to the cardiac cells in the wells.
Ultimately, Eshghi – who specializes in molecular microbiology – says the goal is to work with UHN counterparts, collect antibiotic-resistant bacteria from patients with BE, and use them to discover new inhibitors that prevent modern bacteria from attaching to heart valves.
Power of collaboration
This work is a three-way collaboration via Ted Rogers Centre partners UHN and U of T. The Craig Simmons laboratory in the Centre’s Translational Biology and Engineering Program creates the microplates. Eshghi works in Tara Moriarty’s lab at U of T, which specializes in infectious diseases and thus is experienced with how bacteria attach under shear force. Bacterial strains come from Dr. Andrew Simor’s lab at Sunnybrook.
“When science is done right, it’s always a collaborative effort,” Eshghi says. “We are so specialized in what we do that we can’t truly answer meaningful biological questions without the expertise of multiple individuals.”
He says the Rogers family gift has the power to push research forward.
“This is exactly what everyone talks about in terms of a social commitment, but few ever do. In terms of this Education Fund, it’s huge for someone like me, allowing me to bring my knowledge into a collaborative project. I want to make a meaningful contribution to science, to make an impact on people’s health.”
Eshghi’s co-supervisors on this project are Professors Tara Moriarty and Craig Simmons.